Kidney disease affects millions worldwide, but gaps remain in how we slow disease progression, reduce cardiovascular risk, and improve patient-centric outcomes across every stage. As one of the world's largest academic research organizations with full-service CRO capabilities, the DCRI bridges these gaps through rigorous clinical research, bringing together practicing nephrologists, key opinion leaders, and innovative trial designs to answer the questions that matter most to patients and clinicians.
Chronic kidney disease and kidney failure present unique research challenges: high rates of cardiovascular complications, complex patient populations with multiple comorbidities, persistent health access challenges, and the need for evidence that translates to a range of real-world settings.
The DCRI nephrology and kidney health research program addresses these challenges head-on by ensuring optimal study design, meaningful endpoints, and timely patient recruitment. By combining pioneering, innovative approaches with established relationships with key opinion leaders, we help industry sponsors, federal agencies, and partners advance targeted therapeutics that have transformed the treatment landscape. Regulators recognize DCRI's expertise, providing credibility that sponsors value.
Disease Area Expertise
Our work spans the full spectrum of acute and chronic kidney disease, with particular strength and experience in the following areas:
- Cardiovascular-kidney-metabolic syndrome
- Diabetic kidney disease
- Cardiorenal syndrome
- Obesity and weight loss therapies
- Hypertension
- APOL-1 mediated kidney disease
- Sickle cell nephropathy
- Pharmacogenomics
- Kidney stone disease
- Kidney and dual organ transplantation
- Dialysis and non-dialysis medical management
- Kidney disease in complex comorbidity (e.g., HIV, lupus, end-stage liver disease)
Innovation in Action
DCRI's Nephrology & Kidney Health team is committed to conducting cutting-edge research that advances treatment options and improves patient outcomes. We combine scientific expertise and deep operational experience with innovative methodologies to deliver high-quality, efficient clinical trials.
Our portfolio of innovation includes:
- Leading clinical trial coordinating centers – designed and conducted landmark clinical trials to improve kidney and cardiovascular outcomes in patients with chronic kidney disease.
- Introducing pragmatic elements – integrating real-world approaches into randomized trial design to improve feasibility and generalizability without compromising data quality.
- Leveraging decentralized approaches – utilizing electronic health record data capture and linkage to national databases to ensure comprehensive long-term follow-up, significantly enhancing retention rates and reducing missing data.
- Demonstrating exceptional recruitment of multiple patient populations – partnering with stakeholders and community organizations to ensure study populations are representative of those most impacted.
- Championing implementation trials – increasing the adoption of evidence-based screening and treatment in chronic kidney disease.
- Pioneering innovative data science – applying machine learning, modeling, synthetic control arms, and master protocols to optimize trial efficiency and reduce risk.
Case Study
Seamless Transition of a Phase II to Phase III Trial

Patients with end-stage kidney disease (ESKD) face extraordinarily high cardiovascular risk, yet traditional cardiovascular therapies often fail to translate or have yet to be studied in this vulnerable population. POSIBIL6_ESKD (Prevention of Serious Cardiac Events by Targeting IL-6 in ESKD) tested an innovative monoclonal antibody targeting the IL-6 ligand through a seamlessly integrated Phase II/III program.
Designed as an adaptive program overseen by an industry expert from the DCRI, POSIBIL6_ESKD successfully transitioned two concurrent clinical operations teams across an 8-year, 2,190-patient global program without losing momentum. This strategic approach enabled the DCRI to:
- Execute seamless knowledge transfer from Phase II lessons learned directly to the Phase III team
- Provide real-time site network expansion during the recruitment period
- Make proactive operational adjustments based on emerging data
The Results
- Threefold increase in the number of participating U.S. sites during Phase II recruitment
- Full Phase II enrollment achieved ahead of schedule
- All study milestones were met or exceeded throughout Phase II
- Phase III trial actively enrolling
With POSIBIL6_ESKD, the DCRI showcased our ability to manage complex, long-duration programs in challenging patient populations while maintaining operational excellence and delivering results that exceed sponsor expectations.
Recent News

Largest Study of Its Kind Tests Hydration Strategy for Kidney Stones
Kidney stones can cause some of the most intense pain people ever experience, affecting daily life and leading many to hospital emergency visits. It affects 1 in 11 people in the U.S., and almost half will experience a recurrence. A major new study from the USDRN, coordinated by the DCRI, tested whether a behavioral program could help people drink enough fluids to prevent stones from coming back.
The DCRI Advantage

Clinician-Led Research + Full Service CRO Capabilities
In addition to leveraging the expertise of our practicing physicians and innovative data scientists, the DCRI offers full-service CRO operational capabilities and the unique advantages of being embedded within Duke University's world-class academic medical center infrastructure. With decades of specialized experience in executing complex clinical trials, we are able to reduce study startup timelines compared to traditional CROs. Our proactive operational approach helps us anticipate challenges and deliver effective solutions, ensuring that your trials stay on schedule and within budget.
DCRI’s capabilities include, but are not limited to:
- IRB and regulatory submissions
- Site feasibility, selection, training, and initiation
- Site-, medical-, and safety monitoring
- Full-service electronic data capture and database services
- Data delivery suited for regulatory submissions
- Pharmacovigilance
- Computable phenotypes/EHR-based pre-screening
- Integrated patient recruitment, engagement, and retention solutions
- Advanced analytics and biostatistics experts
Site Network & Partners
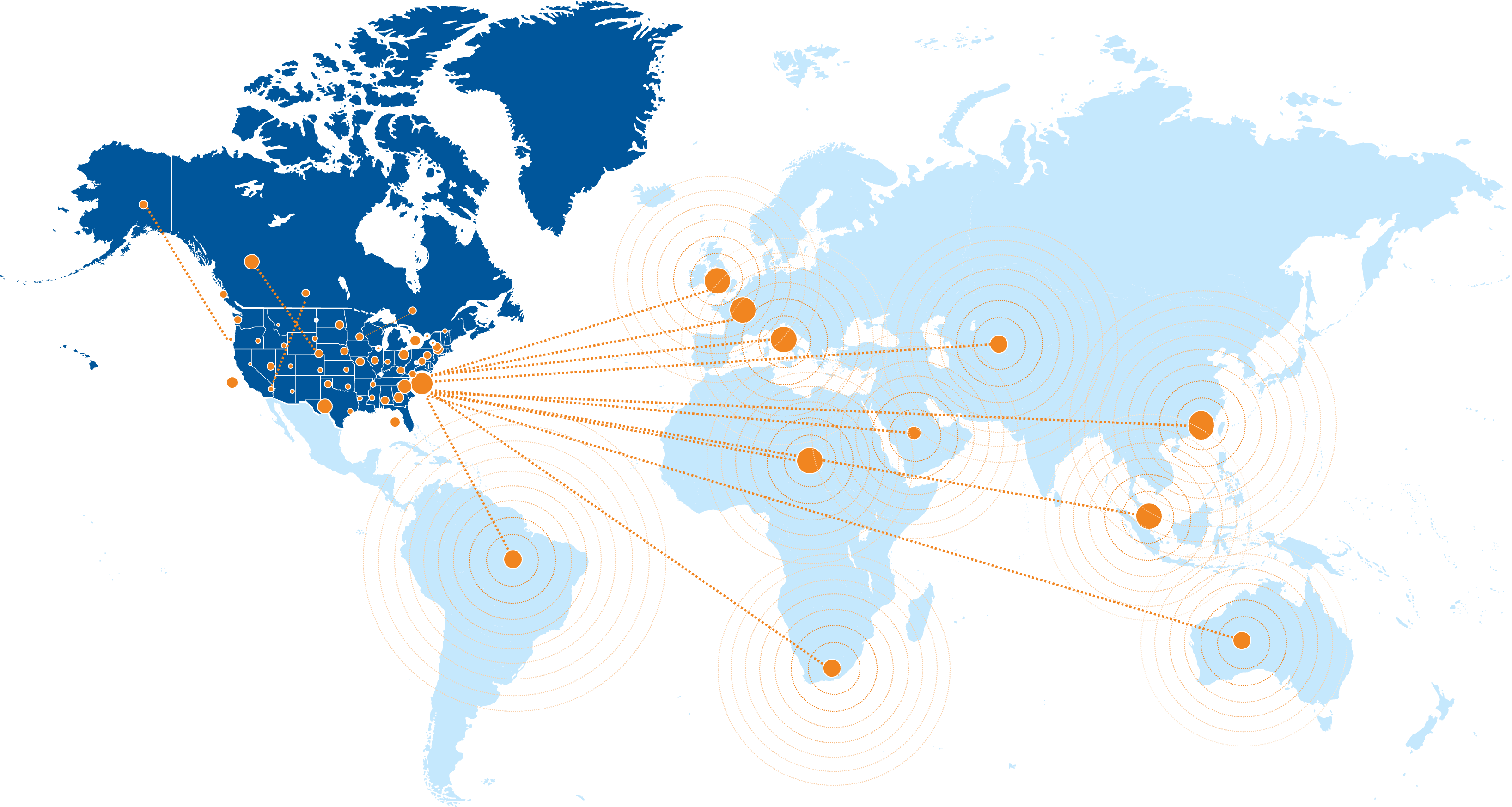
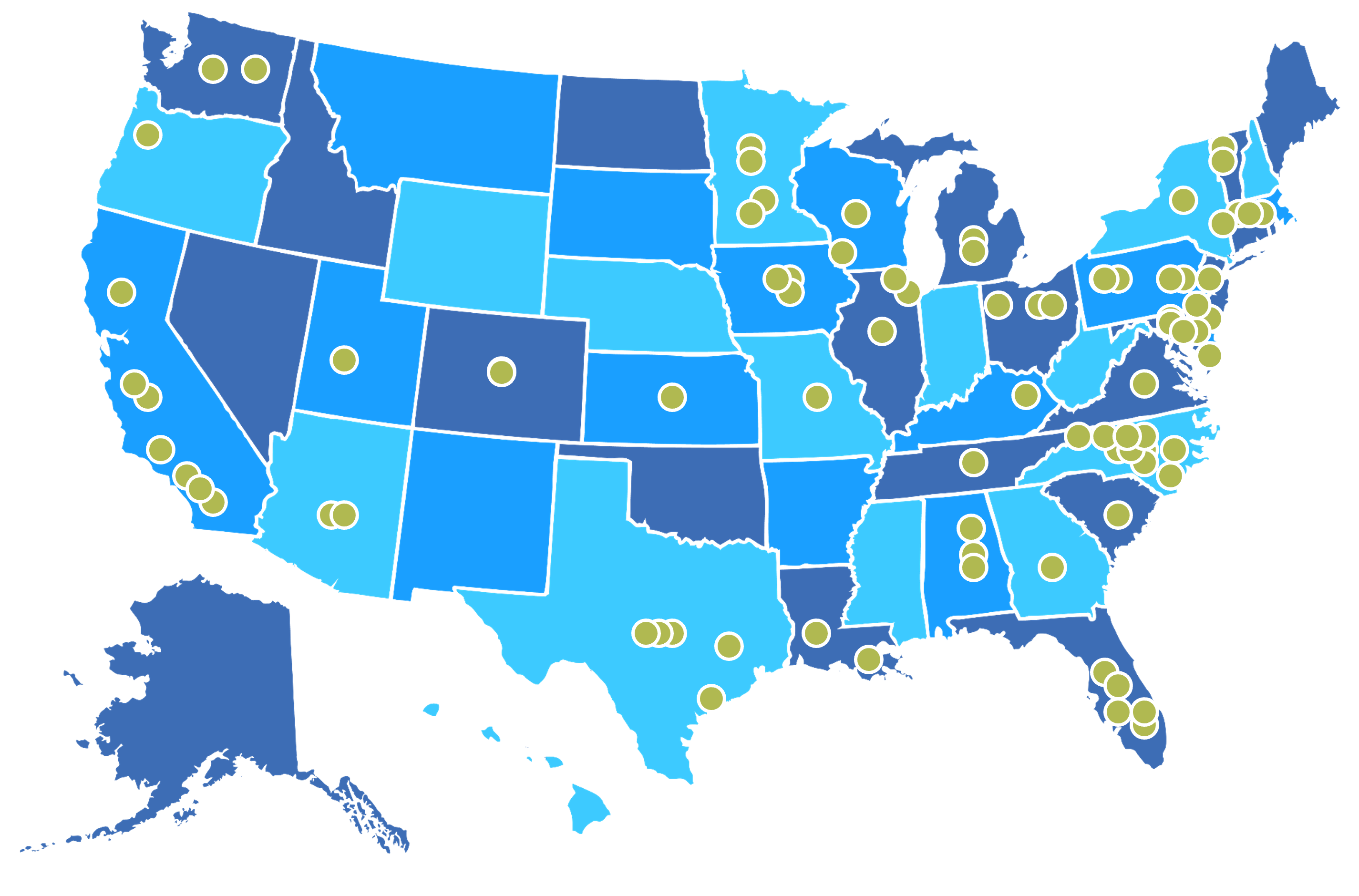
Our trusted network spans over 1,100 research sites across the U.S., Canada, and globally, and includes academic and community-based centers. Through established CRO partnerships, we offer flexible options for conducting studies worldwide, with site selection tailored to specific disease areas and study requirements.
Duke University’s health system is just one of the many sites in our extensive global network. The DCRI Nephrology & Kidney Health team integrates the clinics, resources, and services of this robust ecosystem of expertise, including precision genomics, AI health, population health sciences, and health policy.
Faculty Leadership
The DCRI Nephrology & Kidney Health faculty serve in leadership roles across the field’s most influential organizations and editorial boards, ensuring our research reflects the priorities of the nephrology community and positions the DCRI at the forefront of scientific advances.

Our experts have contributed to peer-reviewed manuscripts and clinical guidelines spanning acute and chronic kidney disease, hypertension, kidney stone disease, dialysis, and kidney transplantation. Collaborating closely with the DCRI operations team, they bring deep knowledge of regulatory pathways and clinical development strategies to each project. As part of Duke’s expansive health system, our faculty also draws on the specialized expertise of the broader Department of Nephrology.
Ready to Advance the Evidence to Improve Kidney Health Together?
Whether you’re developing the next breakthrough therapy or seeking to demonstrate real-world effectiveness, the DCRI Nephrology & Kidney Health team has the expertise, infrastructure, and track record to help you succeed.





