Whether you're planning a global Phase II-IV trial, registry, or a real-world data study, the DCRI combines the operational infrastructure of a full-service CRO with the clinical expertise of Duke University's practicing pulmonary physicians.
Full-Service Respiratory CRO Capabilities, Grounded in Clinical Reality.
Chronic lung diseases affect millions of people around the world, are a leading cause of death, and cause breathlessness, frequent hospitalizations, and impaired quality of life. Despite available treatments, many patients continue to experience a decline in respiratory function, increased disability, and premature death.
The DCRI works to ensure the respiratory therapies and treatments we validate actually reach the patients they are intended to help.
Embedded within Duke University's world-class academic medical center infrastructure, our operational capabilities include:
- Phase II and III global trials
- Phase IV studies and disease registries
- Real-world data analysis, including PCORnet
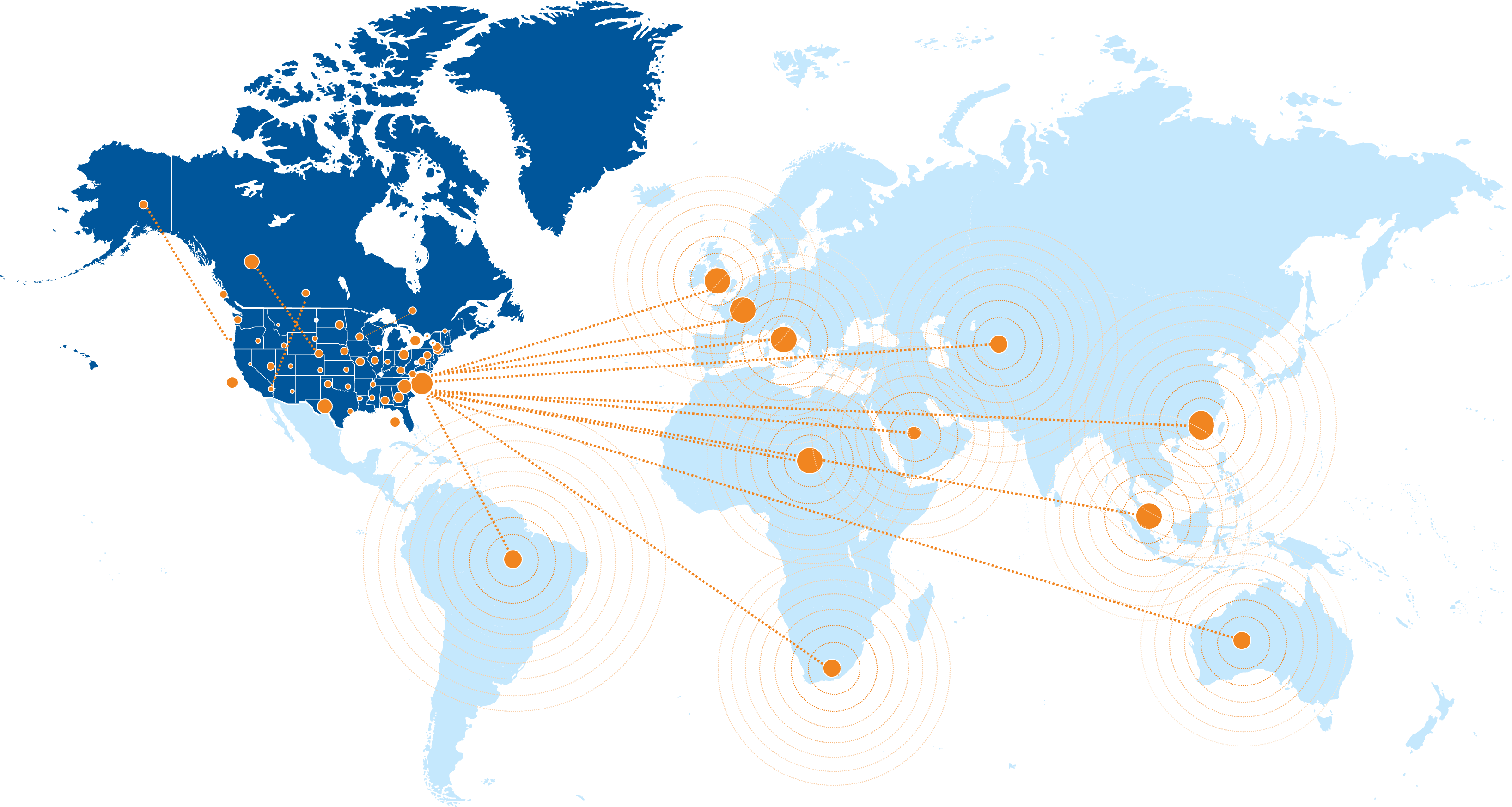
- Network of 1,100 research sites across the United States, as well as established partnerships in more than 40 countries
Trial Expertise That Spans Pulmonary & Critical Care Medicine
- Chronic obstructive pulmonary disease (COPD)
- Interstitial lung disease (ILD) and Idiopathic pulmonary fibrosis (IPF)
- Bronchiectasis
- Asthma and airway biology
- Pneumonia
- Acute respiratory distress syndrome (ARDS)
- Rare lung diseases
- Lung transplantation
- Sleep apnea

Attending the American Thoracic Society's International Conference?
As a Showcase Company, the DCRI is eager to spotlight our evidence-based thought leadership, and meet to discuss how our renowned academic research expertise of the DCRI brings something few CROs can offer.
Academic Excellence, Operationalized.

Rigorous Respiratory Science, Led by the Physicians Who Practice it.
DCRI’s full-service respiratory research capabilities are led by practicing Duke pulmonary physicians who draw on the specialized expertise of the broader Department of Pulmonary, Allergy, and Critical Care team.
- Recognized for their groundbreaking research in asthma, COPD, interstitial lung disease, COVID, ARDS, rare lung diseases, and lung transplantation
- 500+ peer-reviewed manuscripts in the field of respiratory diseases
- Serve on editorial boards of leading respiratory journals
- Principal investigators, steering committee members, and key opinion leaders for major industry trials
We Know Each Pulmonologist Treating our Study Participants
Our trusted network spans over 1,100 research sites across the U.S., Canada, and globally, and includes academic and community-based centers. Through established CRO partnerships, we offer flexible options for conducting studies worldwide, with site selection tailored to specific disease areas and study requirements.
Our Respiratory Research Doesn’t Just Generate Evidence.
It Changes What Physicians Do.
DCRI's respiratory team combines scientific expertise with innovative methodologies to execute studies with meaningful clinical impact. Our portfolio of innovation includes:
- Supporting select U.S.-based sites in AstraZeneca's OBERON Phase 3 trial, while collaborating across a network of community-based and academic research centers to deliver the first clinical evidence demonstrating the benefit of targeting IL-33 in reducing COPD exacerbations.
- Leveraging decentralized approaches in IPF studies to ensure comprehensive long-term follow-up for patients, significantly enhancing retention rates and reducing missing data points critical for regulatory submissions.
- Conducting pivotal clinical trials for IPF that have influenced clinical practice, particularly in advancing lysophosphatidic acid antagonism, demonstrating our ability to execute studies with meaningful clinical impact.
- Demonstrating exceptional recruitment of patients with rare lung conditions, including lung transplants, accelerating timelines in traditionally challenging enrollment scenarios.
- Building strategic partnerships with organizations and CROs to enhance capabilities and support our sponsors' goals, providing flexible, scalable solutions tailored to your specific program needs.
Our innovative, effective, and reliable approach helps sponsors navigate the complex landscape of respiratory medicine development with confidence, delivering the rigorous scientific insights and operational excellence needed to advance your respiratory portfolio.
Disease Spotlight
Interstitial Lung Disease
ILD, including IPF and PPF, remains a challenging therapeutic area with significant unmet medical needs. The DCRI Respiratory Medicine team brings proven expertise across all phases of ILD clinical research:
- Registry Leadership: Our IPF- and ILD-PRO registries have enrolled over 2,200 patients across more than 50 sites, generating real-world evidence on approved therapies, outcomes, and biomarkers for disease progression. Learn more about the registry
- Clinical Trial Execution: Phase II and III trials evaluating novel therapies using both traditional and innovative pragmatic trial designs in collaboration with industry and government partners.
- Data Innovation: Advanced analyses of trial and electronic health record (EHR) datasets to define synthetic control strategies that improve clinical trial efficiency for IPF and other ILDs.
- Real-World Evidence: Collaboration with PCORnet networks to characterize real-world ILD populations and assess disease progression risk using EHRs.
Where Respiratory Research Meets Practice
Whether you're designing a proof-of-concept study or executing a pivotal regulatory trial, the DCRI is where the academic rigor of Duke University meets the operational capabilities of a full-service CRO. Let's move your research forward today.






