
The Duke Clinical Research Institute (DCRI) faculty and operational experts presented or contributed to research across over 70 sessions at the American College of Cardiology Scientific Sessions (ACC.26), held March 28–30 in New Orleans. Their thought leadership encompassed featured presentations, late-breaking clinical trials, moderated debates, digital poster forums, and expert panel discussions as they shared new data, debated emerging evidence, and offered practical guidance on clinical care.
Collectively, their work covered an extensive range of topics across nearly every major cardiovascular subspecialty, including novel trial results, AI-driven innovation, policy, prevention, and the human dimensions of a career in cardiology.
Browse highlights from the DCRI's thought leadership at ACC.26 below, or view the full list of DCRI sessions.
- CHAMPION-AF: Left Atrial Appendage Closure Matches NOACs, Significantly Reduces Bleeding in Landmark AF Trial
- Douglas Calls for Rethinking Heart Attack Prevention
- RECOVER-AUTONOMIC Results Highlight Limits of Heart Rate–Focused Treatment in Long COVID POTS
- A Review of the Ongoing Trials Aiming to Solve ACS's Most Pressing Questions
- Elevated Lp(a) Carries Substantial Economic Burden Beyond Cardiovascular Risk, Simulation Study Finds
- INR Rarely Climbs Above 1.5 in AFib Patients Taking Apixaban, ARISTOTLE Analysis Shows
Sunday, March 29
- Automated Alerts Improve Treatment Rates for Valve Disease in Multicenter Trial
- New Evidence Shows CKD Therapies Offer Profound Cardiovascular Protection
- A Five-Step Recipe for Prescribing GLP-1 Agonists in the Clinic
- Al-Khatib Outlines Path to Personalized Arrhythmia Care Using AI
- Debate Examines Shock Teams vs. Protocols in Cardiogenic Shock Care
- Former FDA Commissioner Calls on Clinicians to Reclaim the Public Health Narrative
Monday, March 30
- DCRI Researchers Introduce Hybrid AI Approach to Streamline Clinical Event Adjudication in Heart Disease Trials
- Experts Unpack New Consensus on Quantitative Coronary Plaque Analysis at ACC.26
- Califf Highlights Federal Policy Impact on Cardiovascular Care
- DCRI Cardiologists Lead Candid Conversation on Parenthood, Balance, and Early Career Life
Saturday, March 28
CHAMPION-AF: Left Atrial Appendage Closure Matches NOACs, Significantly Reduces Bleeding in Landmark AF Trial
During an ACC.26 presentation, it was announced that the Watchman FLEX left atrial appendage closure (LAAC) device was found to be non-inferior to non-vitamin K antagonist oral anticoagulants (NOAC) for preventing stroke and cardiovascular death in patients with atrial fibrillation, while significantly reducing clinically relevant bleeding.
These three-year findings from the CHAMPION-AF trial were published simultaneously in the New England Journal of Medicine. The trial is the largest prospective, randomized, multicenter study comparing LAAC with NOACs, enrolling 3,000 patients across global centers.
Saibal Kar, MD, presented the results on behalf of the trial investigators. Christopher Granger, MD, a faculty member at the DCRI, served on the trial's steering committee, representing a perspective focused on anticoagulation among a group composed largely of device specialists.
Patients enrolled in CHAMPION-AF had non-valvular atrial fibrillation and were considered suitable for long-term anticoagulation. The cohort skewed toward lower risk, with a mean CHA₂DS₂-VASc score of 3.5 and a mean HAS-BLED score of 1.3, which is a clinical tool used to estimate the one-year risk of major bleeding in patients with atrial fibrillation taking anticoagulants. About two-thirds had paroxysmal atrial fibrillation, and nearly half had undergone prior ablation. Participants were randomized 1:1 to receive the Watchman FLEX device or to continue NOAC therapy.
At three years, cardiovascular death, stroke, and systemic embolization, the composite primary efficacy endpoint, occurred in 5.7% of patients in the LAAC group compared with 4.8% in the NOAC group, meeting the pre-specified non-inferiority margin. The primary safety endpoint of non-procedural clinically relevant bleeding was significantly lower in LAAC-treated patients at 10.9% versus 19% (HR 0.55; p<0.001). A net clinical benefit composite endpoint also favored LAAC over NOACs (15.1% vs. 21.8%; HR 0.66; p<0.001). Procedural success was high at 98.8%.
Granger emphasized the results should be interpreted with the study population in mind — a group with relatively lower baseline stroke risk and substantial prior ablation.
"The bottom line was the stroke rates were a little bit higher with LAAC, but in this lower risk group, not substantially higher, and the bleeding rates were lower," he said. "This adds evidence that it may be a reasonable option for many patients."
He emphasized that clinical application would require careful individualization. LAAC appeared especially well-suited for patients with barriers to long-term anticoagulation, he noted.
"It will have to be individualized for patients to see what's the best treatment," Granger said. "This seemed to be a very reasonable approach, especially for those who have a resistance to using anticoagulation."
During the discussion period, the CHAMPION-AF results were contextualized alongside CLOSURE-AF, a recently published trial with somewhat different findings. Discussants noted that CLOSURE-AF enrolled an older, higher-risk population, making direct comparisons challenging and reinforcing the importance of matching treatment to individual patient characteristics.
A five-year endpoint assessing all thromboembolic events will be reported in a future analysis.
Douglas Calls for Rethinking Heart Attack Prevention
Cardiovascular care has made remarkable strides in treating heart attacks, but experts say the next frontier may lie in preventing them altogether and redefining how heart disease is understood.
While participating in a panel discussion, DCRI’s Pamela Douglas, MD, highlighted both the progress and the gaps in current approaches to myocardial infarction (MI), commonly known as a heart attack.

Decades of innovation, including rapid “door-to-balloon” times to restore blood flow, have transformed emergency care and saved countless lives. But Douglas emphasized that advances in acute treatment have not been matched by progress in prevention.
“I think there are a lot of things we don’t know about MI,” she said, pointing to persistent gaps in managing risk factors such as cholesterol and blood pressure, particularly in the years following a heart attack.
Douglas also noted a growing recognition that not all heart attacks are caused by the same underlying mechanism. While traditional models focus on plaque rupture in the arteries, emerging research is identifying other causes, including spontaneous coronary artery dissection, which may require different approaches to diagnosis and treatment.
As a result, she argued, the field may need to move beyond relying primarily on risk factors to assess cardiovascular risk. Instead, she pointed to coronary artery calcium scoring, which directly detects plaque buildup, as a potentially more precise and motivating tool for both clinicians and patients.
“The certainty of disease is more powerful,” Douglas said, noting that clearer evidence of risk can drive behavior change and improve outcomes.
Douglas also highlighted broader systemic challenges, including limited access to preventive tools, gaps in care for rural populations, and underuse of cardiac rehabilitation programs. She suggested that innovations such as virtual rehabilitation and wearable technologies could help expand access and improve long-term care.
Ultimately, Douglas said, achieving the goal of making heart attacks a “never event” will require a more comprehensive approach that combines earlier detection, better risk assessment, and a deeper understanding of the disease’s many forms.
RECOVER-AUTONOMIC Results Highlight Limits of Heart Rate–Focused Treatment in Long COVID POTS
Trial results presented on Saturday afternoon during a late-breaking ACC.26 session offered new insight into treating long COVID postural orthostatic tachycardia syndrome (POTS).
The RECOVER-AUTONOMIC trial evaluated the medication ivabradine in patients with long COVID POTS. The study was funded by the National Institutes of Health’s RECOVER Initiative, with the DCRI serving as the data coordinating center.
POTS is characterized by an excessive increase in heart rate when standing, often accompanied by symptoms such as dizziness, fainting, and palpitations. The condition can significantly affect daily functioning and quality of life, and treatment options remain limited.
In the randomized trial, about 180 participants were assigned to receive either ivabradine or the placebo. Researchers also used a 2-by-2 factorial design to evaluate the impact of “coordinated care,” which included lifestyle guidance, support, and resources alongside standard medical care.

The results showed that ivabradine successfully reduced heart rate compared with placebo. However, the reduction did not translate into a significant improvement in patient-reported symptoms of orthostatic intolerance when the medication was used alone.
A more encouraging signal emerged in a prespecified secondary analysis: patients who received ivabradine in combination with coordinated care experienced improvements in symptom scores compared to those receiving usual care.
The findings reinforce the complexity of post-COVID POTS, which is believed to involve multiple systems in the body, including the autonomic nervous system and immune pathways. Presenter Pam Taub, MD, of the University of California San Diego noted that lowering heart rate alone may be insufficient to address the full range of symptoms.
The study also highlighted the need for more precise clinical endpoints and a better understanding of which patients may benefit most from specific therapies. Future analyses will explore whether certain subgroups respond differently to treatment.
A Review of the Ongoing Trials Aiming to Solve ACS's Most Pressing Questions
Updated acute coronary syndrome (ACS) guidelines have given clinicians a clearer roadmap, but significant questions remain, and a growing portfolio of clinical trials, including one led by the DCRI, is working to fill the gaps.
During a session at ACC.26, DCRI faculty member and interventional cardiologist Jennifer Rymer, MD, MBA, provided an overview of revascularization strategy, mechanical circulatory support, beta-blocker use, and an expanding wave of pharmacologic trials targeting inflammation and platelet biology.
Among the most closely watched is the ARTEMIS trial, run by the DCRI, which completed enrollment in December 2025. The trial is randomizing patients who have experienced a high-risk acute coronary syndrome event to receive either monthly injections of the interleukin-6 (IL-6) inhibitor ziltivekimab or the placebo, with the hypothesis that blunting the early inflammatory surge following myocardial infarction (MI) could reduce infarct size and improve long-term cardiac outcomes. Results are expected by late 2026 or early 2027.
"You may have thought that inflammation died at some point in the past, but it's alive and well," Rymer said.
The interest in inflammatory pathways reflects a broader recognition that myocardial injury after ACS is not purely a mechanical problem. Alongside IL-6 inhibition, NLRP3 inhibitors are also under investigation as upstream anti-inflammatory targets, though one agent in that class recently had its trial terminated after it appeared unlikely to meet its primary efficacy endpoint.
Rymer also highlighted two novel antiplatelet trials that could reshape pre-hospital management of ACS. The CELEBRATE trial, presented at ACC.25, randomized ST-elevation myocardial infarction (STEMI) patients to a subcutaneous glycoprotein (GP) IIb/IIIa inhibitor or placebo, with many patients receiving the injection via ambulance before reaching the catheterization laboratory. The trial demonstrated improved 30-day hierarchical composite outcomes with no significant increase in major bleeding. Separately, SOS-AMI is enrolling about 14,000 high-risk MI patients to test selatogrel, an injectable purinergic receptor (P2Y12) inhibitor that patients self-administer upon recurrence of symptoms. Enrollment will continue through 2026.
On the revascularization front, Rymer noted that the COMPLETE trial, spanning 150 sites across 25 countries and running through 2027, will help clarify whether angiography-guided or physiology-guided approaches to non-culprit vessel percutaneous coronary intervention (PCI) produce better outcomes, particularly in non-ST-elevation myocardial infarction (NSTEMI) populations.
During Rymer's discussion of beta blockers, she cited a randomized controlled trial of approximately 8,000 patients with preserved left ventricular ejection fraction that found no significant benefit on the composite endpoint of death, reinfarction, or hospitalization with chronic beta blocker therapy, prompting her to reconsider her own prescribing practice.
"I have now moved away from chronic use of beta blockers in patients, particularly with preserved ejection fraction," she explained.
The question of when ACS transitions to chronic coronary syndrome, and when de-escalation of antithrombotic therapy is appropriate, also remains unresolved and highly variable in practice, Rymer noted, with ongoing trials examining multiple strategies across a range of patient populations.
Elevated Lp(a) Carries Substantial Economic Burden Beyond Cardiovascular Risk, Simulation Study Finds
Lipoprotein(a) has long been established as an independent, causal risk factor for atherosclerotic cardiovascular disease (ASCVD), but new research presented at ACC.26 suggests that the consequences of elevated Lp(a) generates a substantial, largely unaccounted-for economic toll on patients, caregivers, and society.
The study, presented by DCRI faculty member Nishant Shah, MD, used a Markov model to simulate cardiovascular events and indirect costs across varying Lp(a) levels over five years. Researchers modeled equal-sized groups of 100,000 patients without existing ASCVD, estimating costs from a U.S. societal perspective. The analysis focused on indirect expenses, specifically, patient productivity losses and caregiver-related costs, rather than direct medical expenditures.
The analysis yielded notable results. Compared with patients with Lp(a) below 60 nmol/L, those with levels at or above 150 nmol/L experienced 43.9% more cardiovascular events and incurred 42.4% higher indirect costs. That translated to an incremental burden of $253 million over five years, or about $506 per person per year.
Patients bore the majority of those costs—68.8%. Productivity loss from premature cardiovascular death accounted for 37.9%, followed by disability and early retirement (17.3%), acute and post-event productivity loss (9.6%), and forgone leisure time (3.9%). Caregivers carried the remaining 31.2% through unpaid caregiving time (17.3%), income loss (11.7%), and paid caregiving services (2.2%).
Shah emphasized that the findings have direct clinical implications and that clinicians do not have to wait for next-generation therapies to act. He stressed the importance of educating providers to test Lp(a) at least once in a patient's lifetime, ensuring those with elevated levels receive optimal lipid-lowering therapy, and leveraging existing preventive strategies to reduce cardiovascular risk now.
"It is really important to not only test for Lp(a) early, but treat any risk associated with Lp(a) early in order to be proactive rather than reactive," Shah said.
The study adds meaningful context to a growing conversation about Lp(a)'s population-level impact. It is not solely a biomarker, but also a measurable driver of societal and economic costs. As research into targeted Lp(a) therapies advances, the authors argue that early identification and proactive management will be central to reducing the downstream burden of this undertreated risk factor.
INR Rarely Climbs Above 1.5 in AFib Patients Taking Apixaban, ARISTOTLE Analysis Shows
When a patient with atrial fibrillation (AF) on apixaban turns up with an elevated international normalized ratio (INR), the medication is often the first suspect.
But new data from the ARISTOTLE trial presented at ACC.26 by DCRI fellow Seamus Hughes, MD, suggest that in most cases, clinicians should look elsewhere.
ARISTOTLE is one of the most robust randomized trial datasets in the field of anticoagulation, and was a landmark double-blind, double-dummy randomized controlled trial comparing apixaban to warfarin in patients with AF. This analysis drew from the data that provided monthly INR measurements for all participants, including those assigned to the apixaban arm. That design created a unique opportunity: a large, prospectively collected INR dataset from patients not on warfarin.
The analysis included 8,642 patients assigned to apixaban who had at least one INR measurement, with an average of 4.5 measurements per patient. Researchers used the average INR value from months two through six to characterize each patient's typical INR on the drug.
The median INR was 1.18, with an interquartile range of 1.08 to 1.28. Only 5% of patients, roughly 1 in 20, had a mean INR at or above 1.46. Patients were divided into tertiles: the lowest group had INR values at or below 1.1, while the highest tertile exceeded 1.23.
To understand what drove higher INR values in the apixaban group, researchers used a multivariable proportional odds model. The top five predictors of being in the highest INR tertile were higher hemoglobin, use of the 5 mg apixaban dose compared with 2.5 mg, higher bilirubin, older age, and male sex, factors reflecting patient physiology and hepatic function rather than anticoagulant effect.
In patients on apixaban who present with INR values above 1.5, Hughes said, clinicians should prioritize evaluation for non-apixaban causes of INR elevation — such as hepatic dysfunction, dietary changes, or interacting medications — rather than attributing the finding to the drug itself.
Discussion during the session touched on the potential for real-world data to extend these findings, particularly in exploring INR as a marker for outcomes beyond bleeding.
Sunday, March 29
Automated Alerts Improve Treatment Rates for Valve Disease in Multicenter Trial
A digital alert system integrated into electronic health records may help address persistent undertreatment of serious heart valve conditions, according to findings presented during a late-breaking session on Sunday afternoon.
Results from the ALERT trial, presented by Wayne Batchelor, MD, and simultaneously published in the Journal of the American College of Cardiology, evaluated whether automated clinician notifications could improve care for patients with severe aortic stenosis and mitral regurgitation. The multicenter, cluster-randomized trial included five U.S. health systems and 35 hospitals. DCRI’s Sreekanth Vemulapalli, MD, was part of the study leadership.
Despite effective therapies, fewer than half of eligible patients currently receive timely intervention for valve disease, with disparities across sex, race, and socioeconomic status. The study found that electronic alerts identifying high-risk patients increased referrals to multidisciplinary heart teams and rates of valve intervention within 90 days.
Patients in the alert group were more likely to receive specialty evaluation and treatment and experienced shorter times to care compared with usual practice.
The findings suggest that automated alerts may offer a scalable strategy to improve guideline-directed care and reduce disparities in the treatment of valve disease, particularly by prompting earlier recognition and referral for specialized evaluation.
New Evidence Shows CKD Therapies Offer Profound Cardiovascular Protection
For most patients with chronic kidney disease (CKD), the fear of dialysis looms largest, but that fear may be misplaced, at least when weighed against the cardiovascular threat that it quietly carries.
Speaking at ACC.26 during a session examining novel cardiometabolic health therapies, DCRI nephrologist and faculty member Daniel Edmonston, MD, argued that cardiovascular disease and death, not kidney failure, are the main risks for most CKD patients. This applies regardless of the disease stage.

"The risk of cardiovascular disease and cardiovascular death far outweighs the risk of a kidney event," Edmonston said. "Managing cardiovascular risk is always first and foremost in how we're treating our patients with kidney disease."
To illustrate the magnitude of the disparity, he pointed to data showing that a 35-year-old on dialysis still has a greater risk of cardiovascular death than an 84-year-old without kidney disease. Edmonston described this as a "massive gap" that persists in the field.
That gap deepens as CKD progresses. Cardiovascular mortality risk rises as kidney function declines. It also increases independently with a rising urine albumin-to-creatinine ratio (UACR). These are two compounding risk factors that demand clinical attention. Edmonston emphasized that glomerular filtration rate is usually checked automatically within health systems. In contrast, UACR requires a specific order. Any level of albuminuria, he noted, places a patient on the cardiometabolic-renal spectrum and within reach of therapies that can reduce cardiovascular risk.
The disease phenotype itself shifts with CKD severity. It moves away from mainly atherosclerotic cardiovascular disease and shifts toward left ventricular hypertrophy, pulmonary hypertension, arrhythmias, and heart failure. These changes are driven in part by CKD-specific risk factors. These include uremic metabolic disorders, anemia, iron deficiency, inflammation, and volume overload. These factors compound with traditional cardiovascular risk factors.
Edmonston reviewed the cardiovascular evidence supporting four treatment pillars used to slow CKD progression: renin-angiotensin system (RAS) inhibitors, sodium-glucose cotransporter-2 (SGLT2) inhibitors, nonsteroidal mineralocorticoid receptor antagonists such as finerenone, and glucagon-like peptide-1 (GLP-1) receptor agonists. He argued that each class provides meaningful cardiovascular benefits beyond its renal effects, especially in reducing heart failure risk and major adverse cardiovascular events.
He highlighted the strength of SGLT2 inhibitor data in the CKD population, calling the heart failure risk reduction "profound." He also described the heart failure reduction in the FLOW trial for GLP-1 receptor agonists as a notable surprise. Emerging observational data from the U.S. Renal Data System also suggest cardiovascular benefits for GLP-1 agents in dialysis patients. This population has been historically excluded from randomized trials. Edmonston noted these findings await confirmation in a clinical trial setting.
Many moderate-risk CKD patients will never see a nephrologist, Edmonston told the cardiovascular specialists in the audience. This fact makes the cardiovascular specialist’s role in identifying and managing CKD-related risk even more important.
"CKD, especially advanced CKD, is not just a cardiovascular disease risk factor. At advanced stages, it is a risk equivalent," Edmonston explained.
He noted that for many patients, treating the kidneys means treating the heart.

A Five-Step Recipe for Prescribing GLP-1 Agonists in the Clinic
Preventive cardiologist and DCRI faculty member Nishant Shah, MD, offered a five-step practical framework, framed as a recipe, to help clinicians at ACC.26 navigate the real-world challenges of prescribing glucagon-like peptide-1 (GLP-1) receptor agonists in any clinical setting.
Getting these therapies into the hands of patients who need them most requires more than a prescription pad—it requires a system, he stressed.
"These are cardiovascular drugs," Shah told the audience. "Let's view them as cardiovascular drugs."
The first step is knowing the indication. Approved uses include:
- Type 2 diabetes with or without atherosclerotic cardiovascular disease (ASCVD)
- Chronic kidney disease with diabetes
- Overweight or obese patients with established ASCVD, as supported by the SELECT trial
- Obesity with a BMI of 27 or higher, with at least one comorbidity, or a BMI of 30 or higher
- Metabolic dysfunction-associated steatohepatitis with moderate-to-advanced liver fibrosis
- Moderate-to-severe sleep apnea
Pediatric indications also now exist. Shah emphasized that clearly documenting the indication is essential for navigating prior authorization.
The second step is shared decision-making. Patients should understand the potential for prolonged insurance challenges, early gastrointestinal side effects, and the risk of lean muscle mass loss, which resistance training can help mitigate. Clinicians should also review contraindications, including personal or family history of medullary thyroid cancer or multiple endocrine neoplasia type 2, as well as a history of pancreatitis. Equally important, Shah said, is transparency about cost and access.
"Be honest about coverage in access limitations, so patients aren't surprised that this is a struggle," he said.
The third step is to start low and titrate slowly, particularly in GLP-1-naive patients. Shah noted that in clinical practice, patients may not reach the maximum doses studied in trials but can still achieve meaningful cardiometabolic benefit. When switching agents, he recommended waiting until day seven and starting at a comparable or slightly lower dose; after a prolonged drug holiday, restarting from the beginning. Clinicians should also monitor concomitant medications, including antihypertensives and diabetes therapies that may require adjustment as patients lose weight.
The fourth step is knowing the resources available. At Duke, Shah's team relies on a multidisciplinary model in which a clinical pharmacist leads initiation and titration. For clinicians in community settings without the same infrastructure, he encouraged building whatever system works, including leveraging nurses, advanced practice providers, and pharmacists to create consistent workflows and maintaining a plan for managing prior authorization denials.
The fifth and final step is education and advocacy. Shah urged clinicians to share what works through internal talks and community engagement, and to advocate with policymakers. Coverage for these agents continues to evolve, he noted, and persistence pays off.
"It takes a village," he said. "Don't feel bad about asking for help. The more you prescribe, the more comfortable you'll become."

Al-Khatib Outlines Path to Personalized Arrhythmia Care Using AI
Artificial intelligence has the potential to transform how clinicians detect and treat heart rhythm disorders, but DCRI’s Sana Al-Khatib, MD, MHS, emphasized at ACC.26 that its success will depend on how thoughtfully it is implemented in clinical care.
Al-Khatib described how AI could enable more personalized approaches to arrhythmia care, from predicting sudden cardiac death risk to improving diagnosis, monitoring, and treatment selection.
“The output of any AI largely depends on the input,” Al-Khatib said, emphasizing the importance of high-quality, integrated data.
In cardiac electrophysiology, AI tools are already being explored to analyze electrocardiograms, monitor implantable cardiac devices, and identify patients who may benefit from specific interventions. Some studies have shown that combining multiple data sources like electronic health records, imaging, wearable devices, and implantable monitors can improve the prediction of cardiac events and guide treatment decisions more effectively than traditional methods alone.
Al-Khatib highlighted emerging applications, including the use of AI to predict arrhythmias shortly before they occur, reduce unnecessary device alerts, and improve response to therapies such as cardiac resynchronization therapy. These advances could enhance both clinical outcomes and quality of life for patients.
However, she cautioned that translating promising models into real-world care remains a challenge. Successful implementation requires collaboration across clinicians, data scientists, health systems, and industry partners, as well as careful attention to ethical concerns, data equity, and patient access.
She also emphasized the need for clinical validation, including randomized trials, to determine which AI tools should be adopted in practice. Regulatory pathways and data-sharing limitations remain additional hurdles.
Ultimately, Al-Khatib said, the goal is not to replace clinicians but to support them. Realizing the full potential of artificial intelligence in cardiology will require building systems that are accurate, practical, and trusted, ensuring that innovation leads to meaningful improvements in patient care.
Debate Examines Shock Teams vs. Protocols in Cardiogenic Shock Care
A Sunday morning debate highlighted a key question in cardiogenic shock care: do specialized shock teams or standardized protocols play the bigger role in improving survival?
DCRI’s Jennifer Rymer, MD, argued that shock teams have been associated with improved outcomes, pointing to observational studies showing lower mortality and more consistent use of advanced monitoring and therapies at hospitals that have implemented them. Shock teams bring together specialists across disciplines to make rapid, coordinated decisions for critically ill patients.
At the same time, Rymer acknowledged that protocols designed to reduce variability are also essential.
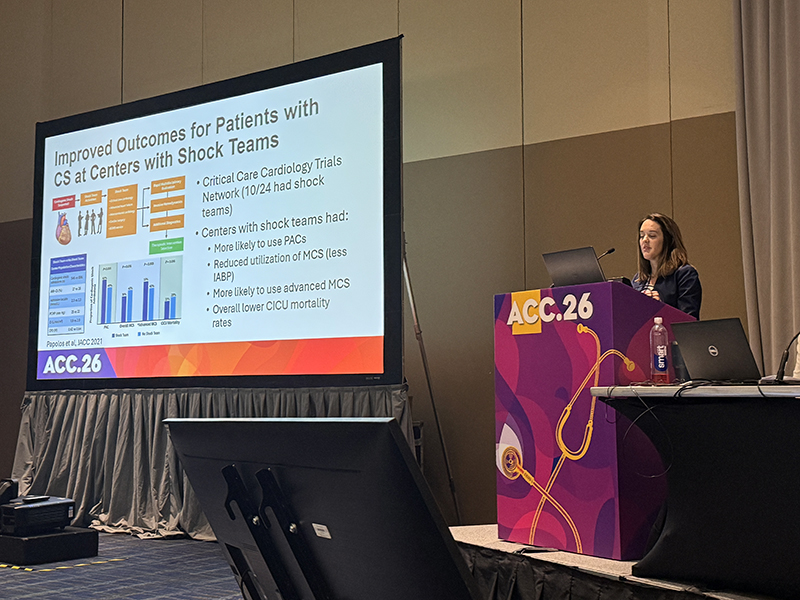
“A good shock team has to work hand-in-hand with a good shock protocol,” she said, emphasizing that protocols are most effective when clinicians are aligned in how they are used.
Her debate opponent, Sandeep Nathan, MD, MSc, of the University of Chicago, argued in favor of protocol-driven care. He highlighted the practical challenges of assembling a full shock team in all settings, particularly at smaller hospitals or during off-hours, and noted that protocols can help standardize care when resources are limited.
Nathan also pointed out that much of the evidence supporting shock teams comes from observational studies, which can be influenced by factors such as increased attention to care or broader improvements over time.
Rymer countered that protocols alone may not achieve their intended effect without strong clinical engagement. Drawing on her experience, she noted that variability can persist if clinicians do not fully adopt or consistently follow protocols.
Both speakers ultimately agreed that the approaches are complementary rather than competing. The discussion underscored a broader challenge in cardiology: translating evidence into practice in a way that is both consistent and adaptable across different care settings.
Former FDA Commissioner Calls on Clinicians to Reclaim the Public Health Narrative
The battle for public trust in medicine is not being lost in clinical journals or on the conference floor. It is being lost at home, at work, and on social media feeds, where most Americans spend the vast majority of their time—and where health professionals are largely absent.
That was the central warning from Robert Califf, MD, former commissioner of the U.S. Food and Drug Administration and founding director of the DCRI, who spoke Saturday at ACC.26 as part of a session titled "Invisible Influences: How Environment, Diet, and Misinformation Shape Heart Health."

"If we don't participate on these platforms, we will lose a battle without ever engaging the competition," Califf said.
Califf framed the problem as both structural and cultural. Despite spending more than $5 trillion annually on health care, the United States has a life expectancy that is 4 to 5 years shorter than that of other high-income nations. This gap, he said, reflects an overinvestment in treating illness and a chronic underinvestment in the conditions that drive it. That documented failure, he argued, has created fertile ground for misinformation and anti-science narratives to take hold.
The information challenge itself is asymmetric. A false health claim can reach a billion people online in seconds. Debunking it requires rigorous work, careful sourcing, and precise communication, placing clinicians at a structural disadvantage from the outset.
Equally important, Califf said, is the way medical professionals have been trained to communicate. The institutional formality and measured detachment that serve experts well in peer-reviewed settings can actively undermine them on the platforms where health misinformation spreads fastest. Success in those spaces, he argued, requires authenticity, expressed vulnerability, and a willingness to engage on the public's terms.
Yet polling data offered a meaningful note of optimism. According to a recent Annenberg Science and Public Health survey, primary care providers remain the most trusted source of health information among the American public, ranking well above federal agencies, national organizations, and public figures. That trust, Califf said, is both an asset and an obligation.
"I would rather have the opinion of an expert than someone who just thought about it 10 seconds ago on a podcast or is trying to sell me an unregulated dietary supplement," he said.
To harness that trust at scale, Califf announced the launch of Coethia, a newly funded, federated organization that brings together health care professionals and biomedical scientists to amplify reliable, evidence-based health information. The name draws on the Greek word "Alethia," meaning truth, combined with "co" to reflect its collaborative design.
Monday, March 30
DCRI Researchers Introduce Hybrid AI Approach to Streamline Clinical Event Adjudication in Heart Disease Trials
ADAPT-CEC, developed by researchers at the DCRI, was showcased at ACC.26 as a novel adaptive artificial intelligence algorithm that demonstrated the ability to generalize across cardiovascular clinical trials, endpoint definitions, and patient populations.
The results of the study, presented by DCRI cardiologist Sreekanth Vemulapalli, MD, raise the prospect of faster, less costly clinical event adjudication in large-scale cardiovascular outcomes research. The findings were simultaneously published in Circulation.
Clinical event adjudication, the independent, expert-driven review of potential cardiovascular events in clinical trials, is considered a regulatory gold standard but is widely recognized as a bottleneck. It is labor-intensive, expensive, and subject to variability across adjudication groups.
The DCRI team trained ADAPT-CEC on data from the ODYSSEY OUTCOMES trial, a multinational study of about 19,000 patients with recent acute coronary syndrome randomized to alirocumab or placebo. Rather than adjudicating directly, a large language model (LLM) first summarized each event dossier, including electronic health records and electrocardiograms, according to a protocol-specified endpoint definition. That structured summary was then processed by an encoder and an in-context meta-learner to generate a probability that a clinical event had occurred.
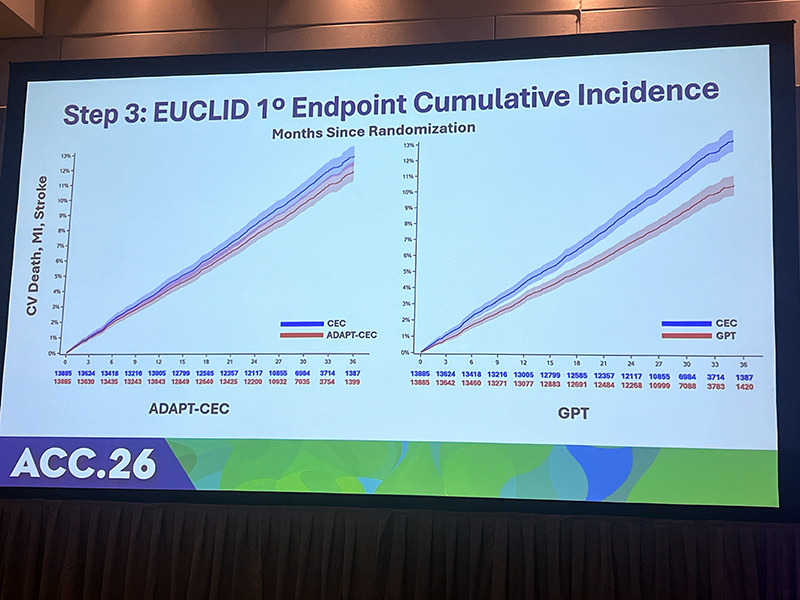
Researchers then tested ADAPT-CEC in the EUCLID trial, a study of about 14,000 patients with peripheral artery disease that compared ticagrelor and clopidogrel and used entirely different endpoint definitions. With as few as 10 example events and 10 non-events per new endpoint, the model successfully adapted to adjudicate stroke, cardiovascular death, and bleeding, which were endpoints on which it had not been trained.
ADAPT-CEC outperformed Chat GPT/GPT-4 alone in adjudicating myocardial infarction (MI) (F1 score: 0.73 vs. 0.697). A hybrid strategy in which the algorithm handled the majority of cases and referred its least certain 30% to human adjudicators yielded the strongest performance across all event types, including the ability to differentiate Type 1 from Type 2 myocardial infarction.
Critically, both ADAPT-CEC and the hybrid model reproduced the EUCLID primary composite endpoints of cardiovascular death, MI, and stroke, with Kaplan-Meier curves nearly overlapping those generated by human adjudicators and with overlapping confidence intervals. GPT-4 alone showed meaningful divergence from human committee results.
"A single AI algorithm trained on a single trial after a brief adaptation can be used to identify the same events with a new definition in a new trial, or even to identify new events," Vemulapalli said.
Discussion by the expert panel following the presentation explored the potential for integrating ADAPT-CEC with electronic health record data at the site level, which could generate initial adjudications for human review and expand the feasibility of future outcomes trials.
Vemulapalli noted that human adjudicators themselves show a 10-20% disagreement rate on re-adjudication—a target the hybrid model aims to achieve.
Limitations include the retrospective nature of the analyses, performance variability across event types, and the fact that training and validation were conducted at a single academic research organization. Prospective evaluation will be required before implementation in active trials.
Experts Unpack New Consensus on Quantitative Coronary Plaque Analysis at ACC.26
With artificial intelligence-powered tools for coronary plaque assessment proliferating faster than clinical guidance can keep pace, a panel of cardiovascular imaging experts gathered at ACC.26 to discuss the landmark 2025 ACC Scientific Statement on Quantitative Coronary Plaque Analysis (QCPA) and to offer practical guidance on when and how these tools belong in clinical practice.
The session was co-chaired by Y.S. Chandrashekhar, MD, editor-in-chief of JACC: Cardiovascular Imaging, and Leslee J. Shaw, PhD of Mount Sinai in New York. The panel also included Jonathon Leipsic, MD, of the University of British Columbia; William R. Herzog, MD, of the U.S. Food and Drug Administration; and Pamela S. Douglas, MD, a DCRI faculty member and contributor to the consensus document. Published in JACC: Cardiovascular Imaging in December 2025, the statement provides the first consensus recommendations on QCPA's clinical use, reporting standards, and future research priorities.
A central take-home message was patient selection. QCPA should be applied when plaque is visually detected on coronary computed tomography angiography (CTA), not for normal scans. Shaw noted that widespread use of QCPA on normal studies in some practice settings was a key driver for establishing this threshold.
Leipsic emphasized where AI-powered QCPA fills gaps in visual interpretation. Namely, the inability to precisely quantify overall plaque burden, characterize calcified versus non-calcified components, or benchmark a patient's atherosclerosis against age- and sex-matched norms.

"The burden of atherosclerosis is the strongest predictor of risk," he said. "The challenge with the human assessment is, I can tell you how many segments of plaque there are, but there can be a little bit of plaque in a segment or a lot of plaque in a segment."
Douglas cautioned that AI output should not be interpreted as standalone thresholds. Risk levels vary by clinical population, sex, and treatment status, and women carry equivalent cardiovascular risk at lower quantitative plaque levels than men. For patients already on statin therapy, she noted that coronary calcium scoring has limited utility and that QCPA or visual segment assessment may better reflect true disease burden.
"It's a clinician that turns that data into information, into knowledge, and then care," Douglas said, emphasizing the human role.
From a regulatory standpoint, Herzog noted the FDA is receiving a growing number of QCPA software submissions and called for a common cross-vendor validation data set, particularly for high-risk features such as low-attenuation plaque. A pre-specified change control plan has been established to streamline review as software continues to evolve.
Coronary calcium scoring retains a Class I recommendation in recently updated lipid guidelines and remains the standard for asymptomatic patients. SCOT-HEART 2, a trial powered to detect cardiovascular events comparing CT-guided to risk score-guided care, is expected to provide more definitive comparative data within about two years.
"We have plenty of evidence. We have great tools," Douglas said. "Our problem is they're not being used, and they're not being used in the right people. CT can help us with that."
Califf Highlights Policy Impact on Cardiovascular Care
Sweeping federal policy changes could have far-reaching consequences for cardiovascular health in the United States, according to experts speaking during a Monday morning ACC.26 session about the impacts of the One Big Beautiful Bill Act.
Presentations during the session outlined evidence on how the recent legislation could affect access to care, health outcomes, and health system stability. Researchers pointed to studies showing that Medicaid work requirements have reduced coverage without increasing employment, while prior Medicaid expansions have been associated with improved access to care and reductions in mortality. Other analyses highlighted the link between food insecurity and cardiovascular disease, with data suggesting that reductions in nutrition assistance could lead to increased hospitalizations among low-income populations.
Speakers also warned that rural health systems, which already face financial and workforce challenges, may be particularly vulnerable to changes in reimbursement and coverage, potentially limiting access to care for high-risk populations.

During the panel discussion, DCRI’s Robert Califf, MD, emphasized that the issues raised extend beyond clinical practice.
“This is a policy problem that calls for policy solutions,” he said, emphasizing that clinicians alone cannot offset the broader structural factors influencing health.
Califf framed the discussion in terms of societal priorities, noting that differences in policy often reflect differing views on individual responsibility versus collective support. He also pointed to systemic inefficiencies, including administrative complexity, as contributors to rising costs without corresponding improvements in outcomes.
Other panelists reinforced the need to address root causes of cardiovascular disease, including hypertension, diet, and access to preventive care. They noted that while advances in medical technology continue, implementation of proven strategies remains inconsistent.

DCRI Cardiologists Lead Candid Conversation on Parenthood, Balance, and Early Career Life
During ACC.26, four parents, who also happen to be experienced cardiologists, engaged in a thought-provoking and honest discussion about the realities of building a family while working in one of medicine's most demanding specialties.
The session was chaired by Jennifer Rymer, MD, a DCRI cardiologist, and included fellow DCRI faculty member Robert Mentz, MD, as well as panelists Nishtha Sareen, MD, of Ascension University, and Geoffrey Barnes, MD, from the University of Michigan Health.
Instead of a prescriptive formula, the group candidly shared personal experiences throughout the hour.
A recurring touchstone was the "80/20 rule": achieving full presence in every role every day is neither realistic nor the goal. The panelists argued that being intentional about where and when you show up matters far more than chasing an impossible standard.
Mentz spoke openly about co-parenting through a divorce and caring for his three children 50% of the time, describing the daily logistics of school drop-offs and the challenge of feeling as if you are constantly underperforming in every role. He shared that he found an unexpected connection with his children in ordinary routines.
"Car time was our super special time," he said. "When your kid is in the back, and they're facing forward, they will share things with you that they won't say any other way."
The panel also tackled professional culture head-on. Sareen described the quiet but persistent pressure many women in cardiology feel to keep parenting invisible at work, including the implication that a spouse caring for children is merely "babysitting." Barnes challenged that framing directly, arguing that the responsibility to normalize family-first limits belongs to everyone.
"I think as men, we have to model that same thing," he said. "That's part of creating the cultural norm."
Sareen, who relocated from Michigan to Nashville for a career opportunity when her daughter was 7, described navigating that transition through honest conversations, new family rituals, and even visits to the cath lab. These moments brought her daughter into her professional world in a tangible way, she said.
The conversation shifted from culture to practicalities: managing call coverage as a proceduralist, handling the high cost of backup childcare, and navigating how to ask colleagues for help while committing to return the favor. The panelists emphasized that reciprocity is not optional.